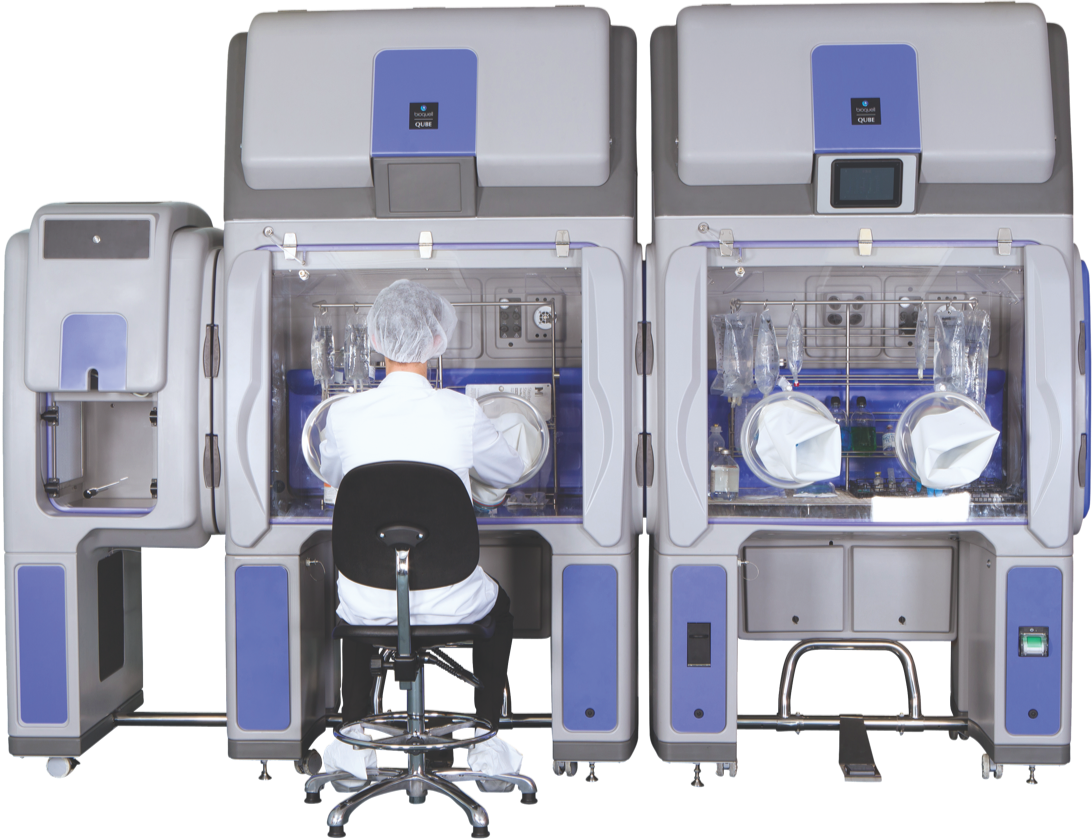
Bioquell Qube
The Bioquell Qube is a configurable isolator integrated with Bioquell’s Hydrogen Peroxide Vapor technology. From its unique design to rapid cycle times, the Bioquell Qube ensures your aseptic workspace needs are met for a safe and productive working environment.
Ideal for:
- Sterility Testing
- Gene & Cell Therapy
- Small Batch Production
- Compounding
- Cytotoxic Preparation
Watch Video 

WHY CHOOSE THE Bioquell Qube

Rapid
Decontamination cycles in as little as 20 minutes; set-up from order to completed installation and validation in as few as 12 weeks

Adaptable
Up to three chambers (two gloves per chamber) and two material transfer areas to meet your workflow needs; positive and negative pressure capabilities

Assured
Customized validated cycles providing
ISO 5/EU Grade A environment; GMP compliance; unidirectional airflow in accordance with GMP Annex 1; 21-CFR Part 11 software available

Efficient
Four levels of environmental monitoring for your viable and nonviable particle needs; ability to incorporate the Merck Millipore Sigma Symbio Flex Sterility Pump; other distinctive options to keep you working efficiently

Integrated
Built-in Bioquell Hydrogen Peroxide Vapor technology for repeatable decontamination results; easy-to-use touch-screen; no HVAC connection required

Productive
Decontamination of materials in the main chamber while you work in another; aseptic-hold retention for typically seven days depending on your protocols

Watch the Bioquell Qube Video
Preview key benefits and features
APPLICABLE SOLUTIONS
Modular and Expandable Configurations
The Bioquell Qube offers diverse applications and options ranging from one to three chambers with up to two optional exterior/material transfers.


The Bioquell Qube for Sterility Testing
Globally accepted with hundreds of chambers currently installed and in use with world leaders in biopharmaceutical, laboratory research and biotechnology, the Bioquell Qube delivers unparalleled efficiency, reliability and ease of implementation to your aseptic environment.
The Bioquell Qube for Cell Therapy
The Bioquell Qube offers a rapid and reliable aseptic environment that can be used in various stages of cell and gene therapy manipulation. See some examples of the Bioquell Qube's impact here.

UNLIKE ANY OTHER ISOLATOR YOU'VE SEEN
See how with the Bioquell Qube infographic.
Download Bioquell Qube InfographicACCESSORIES

Environmental Monitoring
Aseptic environmental conditions with four monitoring modes ranging from manual to fully integrated particle monitoring with both viable and nonviable particle-counting options

Aseptic environmental conditions with four monitoring modes ranging from manual to fully integrated particle monitoring with both viable and nonviable particle-counting options

Up to two integrated Merck Millipore Steritest Symbio Flex sterility test pumps for improving your sterility test process, simplifying operations and freeing up workspace

The patented glove sleeve system remove all creases in both the sleeve and the glove when being decontaminated, and includes smaller and lighter Safe Change Cuff Rings. This design provides the reassurance that all areas of the Bioquell Qube are decontaminated.
Contact us for more information about accessories in the Bioquell Qube to help optimize your workflow.
Contact UsVALIDATION
Our technical experts can install and validate the Bioquell Qube in as quickly as two weeks in most instances.
Because the Bioquell Qube is easy to maneuver, and fast to assemble, you often do not need any construction or electrical alterations to install. It can fit through 800mm (32-inch) door frames to be built directly in the designated area. It requires only a standard electrical outlet to function and, for positive pressure users, does not require connection to HVAC.
Validation includes training, installation, operational and performance qualification (IQ, OQ and PQ), validated cycles based on custom-load configurations, and all appropriate documentation.
FAQs
Will this offer 21-CFR Part 11 compliance?
Yes, we are able to offer an optional data logger for this purpose.
What is the internal floor of the Bioquell Qube made from?
The floor of the Bioquell Qube is constructed from 316 stainless steel to ensure resistance to scratching on the working surface.
I have a small sterility-test need right now. What happens when my needs change?
The Bioquell Qube allows you to add and expand the system as your testing needs change and grow.
How can I bring tubing or cables into the Bioquell Qube?
We have an option for fitting a floor-mounted one-inch triclover port in each chamber. This allows you to bring in liquid lines or cables securely into the body of the Bioquell Qube.
What sets the Bioquell Qube apart from traditional stainless steel options?
The Bioquell Qube is constructed of tough and hard-wearing polypropylene, which is resistant to most materials the unit is ever likely to come into contact with. Because of its construction, the Bioquell Qube can be installed and validated within 12 weeks and, in most cases with minimal invasiveness to your operation or workflow.
Can you set alarms for the temperature inside the Bioquell Qube?
Yes.
If I wish to change pressure from positive to negative, do I need to change the filters or take any additional action to ensure it is safe to use?
The Bioquell Qube can be switched between pressure regimes simply with the touch screen; no additional changes are needed.
Are there different leak tests programmed for the Bioquell Qube?
Yes, there are leak tests programmed for both positive and negative pressure.
Contact Us
To learn more about how Bioquell can fit your solution, please contact us.
The Americas
Bioquell Inc.
702 Electronic Dr. Suite 200
Horsham, PA 19044
+1 215 682 0225
[email protected]










